
XUV is strongly absorbed by most known materials, but it is possible to synthesize multilayer optics that reflect up to about 50% of XUV radiation at normal incidence. The long end of the EUV/XUV spectrum is set by a prominent He + spectral line at 30.4nm. Some other scientific instruments, such as circular dichroism spectrometers, are also commonly nitrogen purged and operate in this spectral region.Įxtreme UV is characterized by a transition in the physics of interaction with matter: wavelengths longer than about 30 nm interact mainly with the chemical valence electrons of matter, while wavelengths shorter than that interact mainly with inner shell electrons and nuclei. By working in oxygen-free gas, the equipment does not have to be built to withstand the pressure differences required to work in a vacuum. This has wide practical significance now that semiconductor manufacturing processes are using wavelengths shorter than 200 nm. Pure nitrogen (less than about 10 ppm oxygen) is transparent to wavelengths in the range of about 150–200 nm. This opacity is due to the strong absorption of light of these wavelengths by oxygen in the air. The onset of vacuum UV, 200 nm, is defined by the fact that ordinary air is opaque below this wavelength. Ordinary window glass passes about 90% of the light above 350 nm, but blocks over 90% of the light below 300 nm. Ordinary glass is partially transparent to UVA but is opaque to shorter wavelengths while Silica or quartz glass, depending on quality, can be transparent even to vacuum UV wavelengths. (Some of the UVB and UVC radiation is responsible for the generation of the ozone layer.) The Sun emits ultraviolet radiation in the UVA, UVB, and UVC bands, but because of absorption in the atmosphere's ozone layer, 99% of the ultraviolet radiation that reaches the Earth's surface is UVA. Many birds have patterns in their plumage that are invisible at usual wavelengths but observable in ultraviolet, and the urine and other secretions of some animals, including dogs, cats, and human beings, is much easier to spot with ultraviolet. Scorpions glow or take on a yellow to green color under UV illumination. Many fruits, flowers, and seeds stand out more strongly from the background in ultraviolet wavelengths as compared to human color vision. Some animals, including birds, reptiles, and insects such as bees, can see into the near ultraviolet. Ultraviolet is colloquially called black light, as it is invisible to the human eye. See 1 E-7 m for a list of objects of comparable sizes. "Vacuum UV" is so named because it is absorbed strongly by air and is used in vacuums.
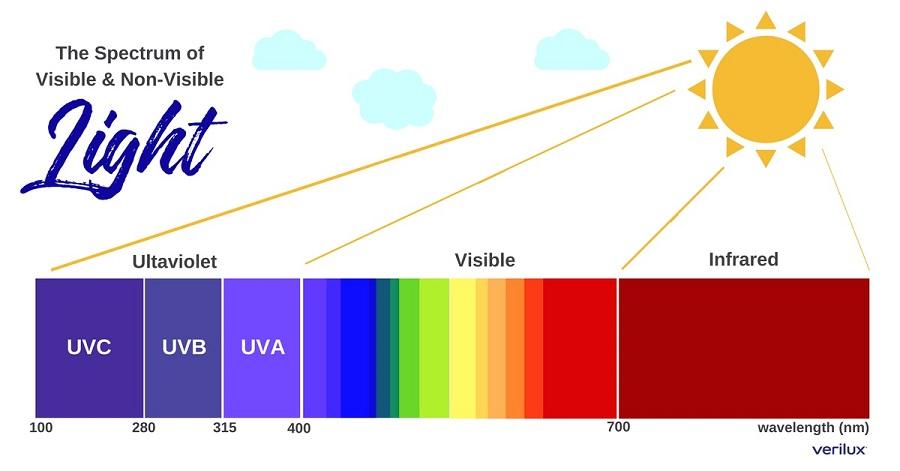
In photolithography, in laser technology, etc., the term deep ultraviolet or DUV refers to wavelengths below 300 nm. The part of the electromagnetic spectrum which ultraviolet light covers can be further subdivided in several different overlapping ways: Name UV light has a shorter wavelength than that of violet light. The name means "beyond violet" (from Latin ultra, "beyond"), violet being the color of the shortest wavelengths of visible light. The terms chemical and heat rays were eventually dropped in favor of ultraviolet and infrared radiation, respectively. The simpler term "chemical rays" was adopted shortly thereafter, and it remained popular throughout the 19th century.

He called them "de-oxidizing rays" to emphasize their chemical reactivity and to distinguish them from "heat rays" at the other end of the visible spectrum. In 1801 the German physicist Johann Wilhelm Ritter made the hallmark observation that invisible rays just beyond the violet end of the visible spectrum were especially effective at darkening silver chloride-soaked paper. The discovery of UV radiation was intimately associated with the observation that silver salts darken when exposed to sunlight. 9.21 Reading completely illegible papyruses.9.20 Preparing low surface energy polymers.9.17 Deterring substance abuse in public places.9.16 Curing of inks, adhesives, varnishes and coatings.7 Degradation of polymers, pigments and dyes.6 Human Health Related Effects of UV Radiation.
